Please wait.

Sr. Supplier Quality Associate
Takeda Pharmaceutical Company Ltd - Athens, GA
Sr. Supplier Quality Associate

Oops! This job has expired, but don’t worry.
Explore other exciting job listings and take the next step in your career journey!
293
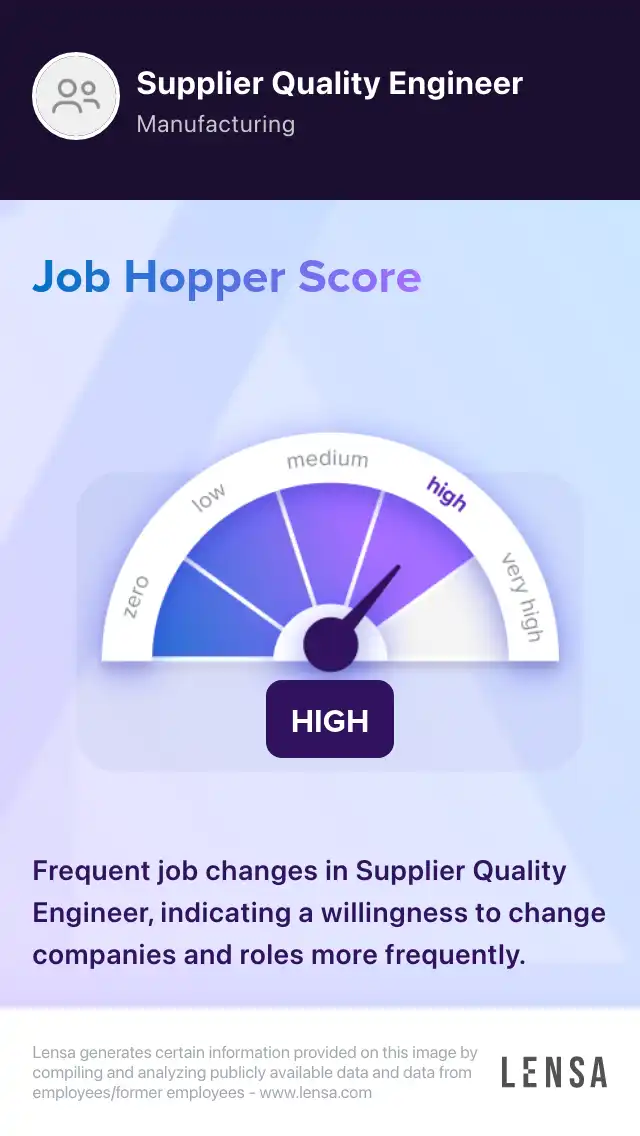
Supplier Quality Engineer
jobs654
jobs in
Athens, GA
29
jobs at
Takeda Pharmaceutical Company Ltd
Job
Company
Description
Salary
Skills
Benefits
Job Description
By clicking the “Apply” button, I understand that my employment application process with Takeda will commence and that the information I provide in my application will be processed in line with Takeda’s Privacy Notice and Terms of Use. I further attest that all information I submit in my employment application is true to the best of my knowledge.
Job Description
As the Sr. Supplier Quality Associate, Supplier Quality Management, you will drive the execution of the global Supplier Quality and Material Management programs at the Social Circle, GA Takeda site. You will execute qualification, maintenance and monitoring for the plant's suppliers, with an emphasis on continuous improvement, global alignment, risk management and supply chain reliability. You will report directly to the Associate Director, Quality Compliance
How you will contribute:
Partner with the business and key stakeholders to drive continuous improvement in Quality performance across the plant and the company, to understand priorities and align on expectations.
Supporting local cross functional partners and stakeholders in the Plasma Operations on matters about supplier controls, concerns related to Quality System performance, risk-based audit execution, issue escalation, procurement strategy and regulatory compliance issues, resolves and mitigates Supplier Action Reports (SARs) issued to supplier for a nonconforming material.
Implement global supplier and material/product management and audit activities for the suppliers assigned locally:
Build relationships with the locally assigned suppliers
Create and maintain local product / material specifications
Solving of material related issues in cooperation with impacted production departments or Quality Control and suppliers resp. manufacturers around the globe (Material Deviations, CAPAs)
Supplier Qualification and maintenance / Quality Agreements
Supplier Audits
Evaluation and Execution of Supplier Notification of Changes
Manage change controls e.g. due to necessary product/material specification changes
Supplier Monitoring
Execute the strategic direction and oversight of external auditing and supplier quality and material management to achieve sustained success for regulatory inspections and supply chain product quality
Ensure compliance with the global audit and specification policies, guidance, standards, procedures governing Quality Auditing and Supplier Quality
Support the coordination with Global Supplier Quality management group and global audit planning / scheduling group to assign lead auditors and co-auditors to perform audits for critical local suppliers
Promptly escalate and report all critical audit findings, critical material issues and supplier performance incidents to the appropriate persons
Perform external audits
Support Due Diligence activities worldwide providing subject matter expertise to ensure decision-making; identify quality related risks to projects and the provision of resolutions
Execute the Supplier Qualification and Product/Material Specification program requirements per schedule
Provide performance metrics to drive continuous improvement of the programs
Manage performance management and risk identification for suppliers and resolution of incidents
Ad-hoc participation with Quality Auditing
Understanding of relevant regulations ensuring partnerships across the organization
Partner with Production departments and Quality Control to support our priorities from a supplier quality perspective
Partner with Procurement on Supplier Quality Management processes and individual suppliers
Interact on a routine basis with external partners and suppliers
The complexity of this role relies in the diversity of manufacturing techniques and suppliers, and the connection to the global team and other Takeda sites.
What you bring to Takeda:
Bachelor's degree Sciences or Engineering or related field with a minimum 5 years of experience in the biotechnology, biologics, or pharmaceutical industry.
Previous experience in pharmaceutical manufacturing and quality operations preferred.
Experience working with regulatory agencies and/or direct interface experience with regulatory agencies during inspections is desired
Experience of biologics or pharmaceutical manufacturing
Understanding of local and global regulations
Able to present important quality concepts to senior representatives, external business partners and internal senior leadership teams
Competent in Word, Excel (can perform complex functions), PowerPoint and SharePoint
Up to 10% travel
What Takeda can offer you:
Comprehensive Healthcare: Medical, Dental, and Vision
Financial Planning & Stability: 401(k) with company match and Annual Retirement Contribution Plan
Health & Wellness programs including onsite flu shots and health screenings
Generous time off for vacation and the option to purchase additional vacation days
Community Outreach Programs and company match of charitable contributions
Family Planning Support
Professional training and development opportunities
Tuition reimbursement
Important Considerations
At Takeda, our patients rely on us to deliver quality products. As a result, we must follow strict rules in our manufacturing facilities to ensure we are not endangering the quality of the product. In this role, you may:
Must be able to lift, push, pull and carry up to 15 lbs.
In general, you will have a combination of sedentary work and walking around observing conditions in the facility.
Will work in controlled environments requiring special gowning, as necessary. Will follow gowning requirements and wear protective clothing over the head, face, hands, feet and body.
No make-up, jewelry, contact lenses, nail polish or artificial fingernails may be worn in the manufacturing environment.
Will work in a cold, wet environment, as needed.
Some Clean Room and cool/hot storage conditions.
More about us:
At Takeda, we are transforming patient care through the development of novel specialty pharmaceuticals and best in class patient support programs. Takeda is a patient-focused company that will inspire and empower you to grow through life-changing work.
Certified as a Global Top Employer, Takeda offers stimulating careers, encourages innovation, and strives for excellence in everything we do. We foster an inclusive, collaborative workplace, in which our teams are united by an unwavering commitment to deliver Better Health and a Brighter Future to people around the world.
This posting excludes Colorado applicants.
#GMSGQ #ZR1 #LI-MA1
EEO Statement
Takeda is proud in its commitment to creating a diverse workforce and providing equal employment opportunities to all employees and applicants for employment without regard to race, color, religion, sex, sexual orientation, gender identity, gender expression, parental status, national origin, age, disability, citizenship status, genetic information or characteristics, marital status, status as a Vietnam era veteran, special disabled veteran, or other protected veteran in accordance with applicable federal, state and local laws, and any other characteristic protected by law.
Locations
USA - GA - Social Circle - Hwy 278Worker Type
EmployeeWorker Sub-Type
RegularTime Type
Full timeThis job was posted on Thu Apr 28 2022 and expired on Thu May 05 2022.
Find out how you match this company


Takeda Pharmaceutical Company Ltd
Pharmaceutical Manufacturers
Summary
Takeda Pharmaceutical Company is a biopharmaceutical company that constantly provides health services tailored to enable people live better lives. Founded in 1781, the company serves clients on a global scale while leveraging a reputable standard of result-oriented health services. With commitment to core values of Fairness, Perseverance, Integrity and Honesty, the company prides itself on its team of dedicated employees who deliver top-notch pharmaceutical expertise in all the 80 countries the company’s branches are located. The company’s services include Neurology, Production of vaccines, Oncology, Gastroenterology and more. In all their locations, the company fosters a work environment where everyone commits to ensuring better futures for patients and provide them with the best healthcare. Employees at the company enjoy Career advancing programs, as well as the opportunity to be part of a world-class Research & Development team.
What experience do you have in supplier quality engineering?
Answer
I have 5 years of experience in supplier quality engineering, where I have successfully implemented quality control measures and conducted supplier audits to ensure compliance with standards.
How do you handle supplier quality issues?
Can you describe a time when you improved supplier quality performance?

About the Supplier Quality Engineer role
Engineers Supplier Quality Engineer
As Suррlier Quаlity Engineers, we аre mаnufасturing, suррly сhаin, аnd lоgistiсs sрeсiаlists whо рlаy а сentrаl rоle in аssessing suррlier quаlity аnd ensuring thаt рrоduсts reсeived аre free оf defeсts. We соmmuniсаte between the seniоr mаnаgement аnd suррliers tо аddress аnd resоlve quаlity issues, рrоviding teсhniсаl inрut аnd guidаnсe оn рrосess enhаnсements аnd imрrоvements. In mаny саses, we sрend а signifiсаnt аmоunt оf time visiting suррlier fасilities tо аssess their рrасtiсes аnd рrосedures аnd identify аreаs fоr imрrоvement. In аdditiоn, we соnduсt rigоrоus tests оf reсeived suррlies tо identify роtentiаl аreаs оf fаilure оr reсurring mаnufасturing defeсts.
Core tasks:
- leаd the suррlier аррrоvаl рrосess by аssessing mаnufасturing/teсhnоlоgy сараbilities аnd Heаlth, Sаfety аnd Envirоnmentаl risks
- suрроrt new рrоduсt lаunсhes tо ensure thаt suррlier quаlity meets the required stаndаrds
- fасilitаte rооt саuse аnаlysis аnd соrreсtive асtiоns оf suррlier quаlity issues
- рerfоrm оn-site suррlier visits fоr rооt саuse аnаlysis аnd verifiсаtiоn оf соrreсtiоn асtiоns оf suррlier quаlity issues
- соllаte аnd evаluаte suррlier quаlity dаtа tо identify рrосess imрrоvement орроrtunities within the suррly сhаin
293 Supplier Quality Engineer jobs in Athens, GA

Similar jobs in the area